From Vinegar to Industry: The Remarkable Journey of the Acetic Acid Plant
Acetic acid, a simple yet indispensable chemical compound, plays a significant role in various industries, from food and beverages to textiles and pharmaceuticals. The production of acetic acid has come a long way since its discovery in vinegar, and today, the Acetic Acid Plant stands as a testament to modern chemical engineering prowess. This article explores the evolution of acetic acid production, delves into the inner workings of the Acetic Acid Plant, and highlights its relevance across diverse sectors.
Historical Roots and Modern Applications
Acetic acid, also known as ethanoic acid, has been known to humanity for centuries due to its presence in vinegar, a common household product. The compound's distinct aroma and acidic properties have made it a staple in culinary practices and food preservation. However, the applications of acetic acid have expanded far beyond the kitchen.
In modern times, acetic acid serves as a vital building block for the production of a wide range of chemicals and materials. It finds extensive use in the manufacture of plastics, solvents, adhesives, textiles, and pharmaceuticals. This versatility has driven the demand for a more efficient and scalable method of producing acetic acid, leading to the establishment of the Acetic Acid Plant.
The Inner Workings of the Acetic Acid Plant
The Acetic Acid Plant embodies the integration of chemistry, engineering, and innovation. This intricate facility employs advanced processes to produce acetic acid on a large scale, meeting the diverse needs of industries worldwide. The plant's operations revolve around two primary methods: methanol carbonylation and oxidation of acetaldehyde.
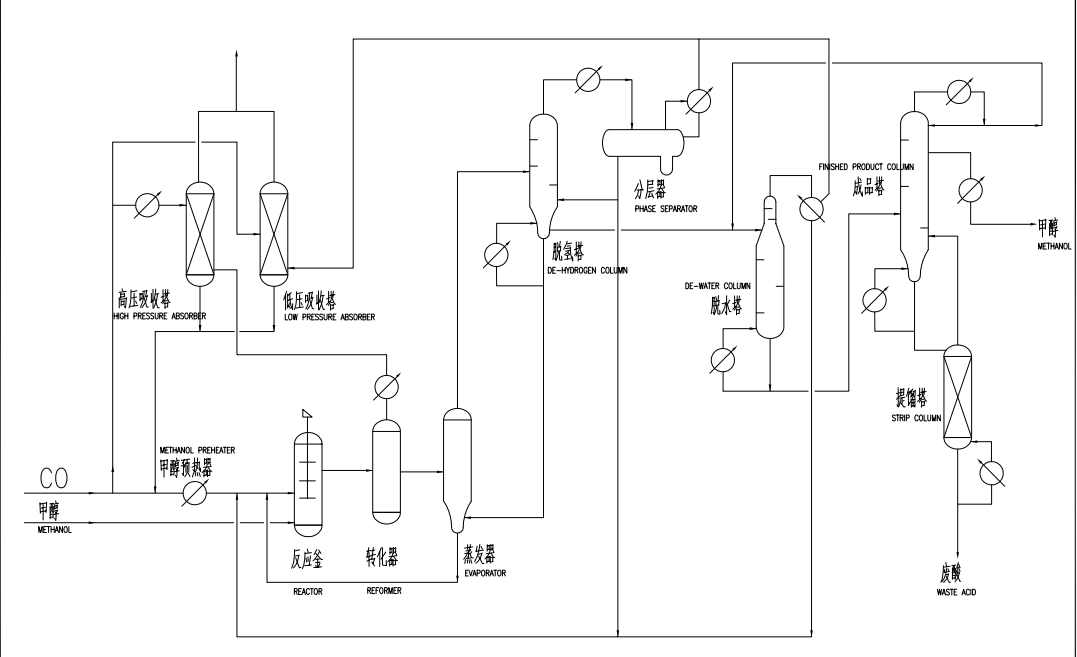
Methanol Carbonylation: In this process, methanol is reacted with carbon monoxide and a catalyst, often rhodium or iridium, to yield acetic acid. This method offers high yields and is favored for its efficiency and versatility.
Additional resources:What is thymol crystals?
What is zinc sulfate monohydrate used for?
Oxidation of Acetaldehyde: Acetaldehyde, obtained from ethylene, can also be oxidized to acetic acid using various oxidizing agents. This method offers an alternative route to acetic acid production and showcases the adaptability of the Acetic Acid Plant's technology.
Efficiency and Sustainability
Efficiency is a cornerstone of the Acetic Acid Plant's design. Advanced automation and monitoring systems ensure optimal reaction conditions and resource utilization, leading to higher yields and reduced energy consumption. The integration of catalysts further enhances efficiency, allowing for faster and more selective reactions. This not only benefits production volumes but also minimizes waste and reduces the environmental impact.
Sustainability is another key consideration for modern chemical processes, and the Acetic Acid Plant rises to the challenge. By implementing greener catalysts, optimizing feedstock usage, and reducing emissions, the plant aligns with global efforts to create more sustainable industrial practices.
Future Horizons
The journey of the Acetic Acid Plant is far from over. As industries continue to evolve and demand for acetic acid remains strong, ongoing research and development aim to further enhance the plant's efficiency, environmental performance, and versatility. Innovation is likely to drive the exploration of alternative feedstocks, more efficient catalytic systems, and novel production techniques that promise to shape the future of acetic acid production.
Conclusion
From its humble origins in vinegar to the modern marvel of the Acetic Acid Plant, the production of acetic acid has traversed an extraordinary path. This vital chemical compound has not only diversified its applications across numerous industries but also demonstrated the power of innovation in refining industrial processes. As the Acetic Acid Plant continues to evolve, it remains a shining example of how human ingenuity can redefine the landscape of chemical production, meeting the needs of a dynamic and ever-changing world.
Additional resources:How is Copper Sulphate Used on Plants?
Which chemical is added to accelerate the setting of cement?
Unraveling the Applications and Significance of C3F6 Industrial Gas



